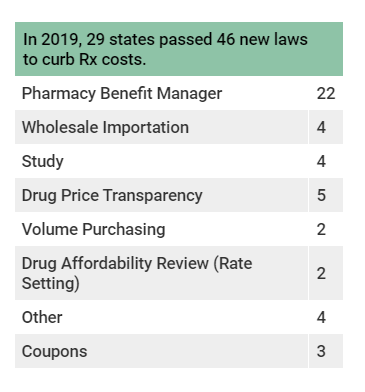
Most state legislatures have adjourned after busy sessions that again focused on reining in prescription drug prices. By the end of June, 29 states had enacted 46 new drug pricing laws that build on earlier work and took more comprehensive and far-reaching approaches to curb rising drug prices.
As in years past, states targeted pharmacy benefit managers (PBMs), but did so more aggressively and sought to extend transparency requirements to all entities in the drug supply chain. Transparency is an important first step – policymakers need to understand the black box of drug pricing to be effective legislators – but transparency by itself does not lower prices. Several states moved beyond transparency:
- Two states created drug affordability review boards;
- Two states approved initiatives to leverage public purchasers to enhance volume purchasing strategies; and
- Several states passed wholesale drug importation programs.
PBMs
Last year, many states took their first steps to regulate opaque PBM practices. The most common action was to ban the use of gag clauses in contracts between PBMs and pharmacies that prohibited pharmacists from disclosing less-costly drug cost information to consumers. This year, states went farther to ensure that PBMs obtain licensure, limit the use of spread pricing, and pass along any manufacturer rebates to consumers or health plans.
Louisiana and Arkansas now have laws that require PBM licensure and prohibit PBMs from conducting spread pricing. Licensure allows state officials to know how many and what entities are operating as PBMs in their state. It also gives the specific state agencies more enforcement authority should PBMs engage in fraudulent activity. Limiting spread pricing ensures that health carriers pay the same amount for a drug that the PBM pays a pharmacy to dispense that medication to a consumer, thereby increasing price transparency.
Minnesota and Maine placed new requirements on PBMs’ use of maximum allowable cost (MAC) lists, which PBMs currently manipulate to charge health plans more for a drug than the amount they reimburse pharmacies for dispensing the drug to a consumer. Under these new laws, PBMs must report which sources are used to determine MAC pricing and provide an appeals process for plans.
The New York legislature also passed a measure requiring PBM licensure and transparency reporting to health plans and the state’s insurance department. The bill stipulates that PBMs have a duty and obligation to not just health plans but to also covered individuals to perform their services with care, skill, prudence, diligence, and professionalism. The measure passed with broad support and now awaits action from Gov. Andrew Cuomo.
Drug Affordability Review Boards
After the Fourth Circuit Court of Appeals stuck down Maryland’s 2017 anti-price-gouging law last year, lawmakers tried a different approach and approved the first-in-the-nation prescription drug affordability board. The original goal of the legislation was to establish upper payment limits (UPL) for certain high-cost drugs in order to protect payers in the state from paying more than that UPL for those drugs. Maryland’s compromise law creates a five-member board that will study the entire pharmaceutical distribution and payment system, as well as policy options used by other states and countries to lower drug prices, including setting UPLs and implementing a bulk purchasing process. This law includes a pathway to setting UPLs for state purchasers should the board find UPLs to be in the best interest of the state. Should that approach be implemented, the state could extend the UPLs to other payers.
In a similar move, Maine also established a drug affordability review board. The Maine board is required to set prescription drug spending targets for public entities and must consider strategies that can help public purchasers meet expenditure goals, including collaborating with other states and allowing carriers who cover small businesses and individuals to buy into a public payer drug benefit plan. The targets are goals, but the board will report annually to the legislature on the progress of public purchasers, whose appropriations must be approved by the legislature, in meeting goals
Leveraging Public Purchasing
During this legislative session, more states expressed interest in cross-agency strategies to leverage their buying power to lower drug costs as a way to reduce spending. California’s governor issued an executive order compelling public purchasers to work together. New Mexico established the Interagency Pharmaceuticals Purchasing Council to review cost-containment strategies for the procurement of drugs and pharmacy benefits. The council must identify ways to leverage constituent agencies’ purchasing power to lower drug spending. It must also study strategies such as establishing a common formulary, using a single purchase agreement or using common procurement of expert services for all constituent agencies. Delaware established a similar Interagency Pharmaceuticals Purchasing Study Group to review and make recommendations to leverage the bulk purchasing power of the state to negotiate lower pharmaceutical prices. Maine’s board similarly requires diverse public purchasers to work collaboratively to lower drug spending.
Importation
This session, Florida, Colorado, and Maine joined Vermont (which approved importation in 2018) to allow their states to design wholesale drug importation programs to purchase select drugs from Canada and seek approval from the US Department of Health and Human Services to implement the program. Each state must create a program that meets federal safety requirements and results in savings for consumers.
Takeaways
The new laws passed this session clearly build on the groundwork and lessons learned from previously implemented state laws. Having passed broad legislation over the past few years, many states pivoted to targeted policies that are poised to have stronger, more specific implications for all entities in the pharmaceutical supply chain. For more information about state action on pharmaceutical costs, read descriptions of all newly-enacted laws and explore NASHP’s Legislative Tracker.